BD PhaSeal™ Optima system
The next-generation Closed-System Drug-Transfer Device (CSTD) from BD that advances hazardous drug protection

- Overview
- Product & Accessories
- eIFU & Resources
Safety. Performance. Ergonomics. Ease of use. We've optimized every component, and the result is a CSTD with design innovations that advance hazardous drug safety. The BD PhaSeal™ Optima system meets the requirements of the NIOSH IPA draft CSTD-test protocol‡, a robust test protocol applicable to barrier-type closed system drug transfer devices.1,2
Notes
* Within an ISO Class V environment following aseptic technique
** 2015
^ The ability to prevent microbial ingress for up to 168 hours should not be interpreted as modifying, extending or superseding a manufacturer's labeling recommendations for the storage and expiration dating of the drug vial. Refer to drug manufacturer's recommendations and USP compounding guidelines for shelf life and sterility information.
References
- Centers for Disease Control and Prevention (CDC). Easy Ergonomics: A Guide to Selecting Non-Powered Hand Tools. https://www.cdc.gov/niosh/docs/2004-164/pdfs/2004-164.pdf. Published August 18, 2004. Accessed September 2, 2020.
- BD Engineering study: Evaluation of PhaSeal Optima residual drug loss performance testing. 2017.
- BD Engineering study: Evaluation of PhaSeal Optima residual drug loss performance testing. 2018.
- Gilbar PJ, Chambers CR, Gilbar EC. Opportunities to significantly reduce expenditure associated with cancer drugs. Future Oncol.2017;13(15):1311-1322.
- McMichael DM, Jefferson DM, Carey ET, et al. Utility of the PhaSeal closed system drug transfer device. Am J Pharm Benefits.2011;3(1):9-16.
- Bendeka [prescribing information], North Wales, Pennsylvania. Teva Pharmaceuticals USA, Inc. 2018.
- Doxil [prescribing information], Bedford, Ohio. ALZA Corporation. 2016.
- Herceptin [prescribing information], South San Francisco, California. Genentech, Inc. 2017.
- Valstar [prescribing information], Chadds Ford, Pennsylvania. Endo Pharmaceuticals, Inc. 2017
- April 2018 ASP pricing file. CMS website. https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Part-B-Drugs/McrPartBDrugAvgSalesPrice/2018ASPFiles.html. Accessed June 8, 2018.
BD-17517 (06/24)
Airtight
Every component is designed with no inlets or air exchange for completely airtight hazardous drug transfers.
Demonstrated
The BD PhaSeal™ Optima system prevents microbial ingress for up to 168 hours and 10 penetrations*†
Efficient
The BD PhaSeal™ Optima system minimises residual drug loss in vials compared to other similar system.
User-validated
Feedback from longstanding CSTD users and traditional technique users guided us to optimise and innovate the design.
Safety
- Injector designed to prevent accidental needlesticks
- Self-sealing membranes for leakproof connections
Performance
- Proprietary protector design to maximise drug extraction
- Compatible with ISO-compliant Luer lock syringes and IV sets
Ergonomics
- Power grip and straight motion to connect and disconnect, aligned with NIOSH recommendations to avoid pinch grip3
- Optimised design for clinician comfort
Ease of use
- Intuitive one-step straight-push connection to aid workflow
- Can connect without alignment to help minimise learning curve
This is a modal window.
Beginning of dialog window. Escape will cancel and close the window.
End of dialog window.
This is a modal window. This modal can be closed by pressing the Escape key or activating the close button.
BD PhaSeal™ Optima Video for Pharmacy
This is a modal window.
Beginning of dialog window. Escape will cancel and close the window.
End of dialog window.
This is a modal window. This modal can be closed by pressing the Escape key or activating the close button.
BD PhaSeal™ Optima for Nursing
-
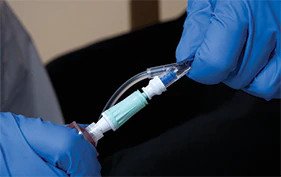
Protect clinicians and patients from exposure to hazardous drugs with a closed system
-

Get results in less than 10 minutes with the first and only rapid hazardous drug* detection system



