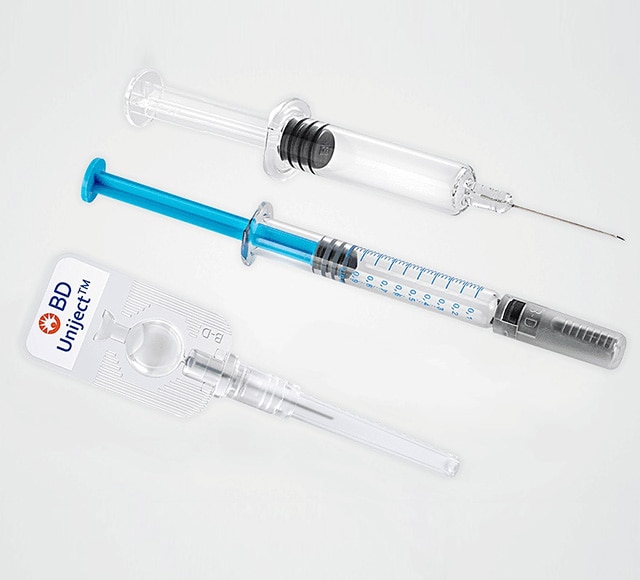
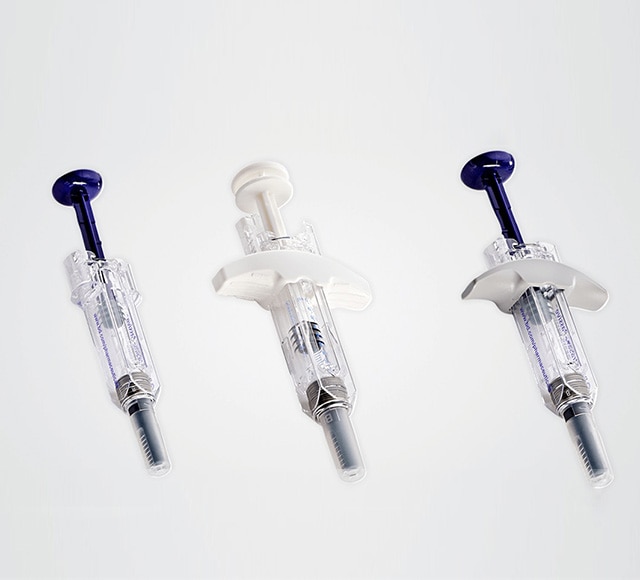
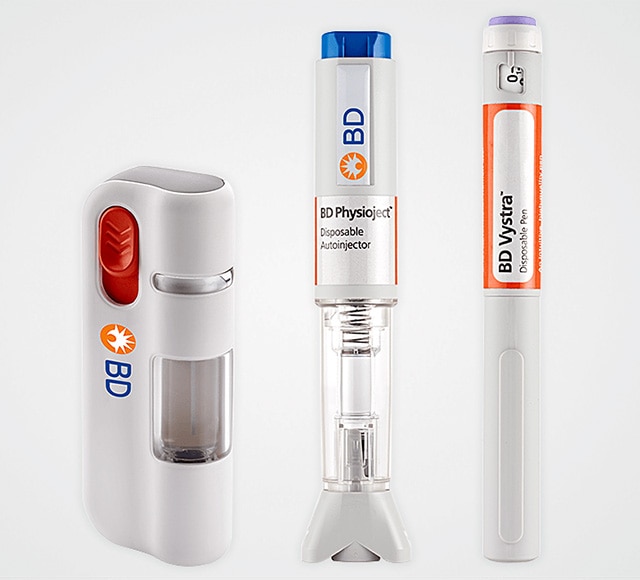
System Integration
Effective system integration can be critical to the success of combination products for pharmaceutical companies.5

As your partner, we leverage our deep industry expertise to help enhance device related aspects of your combination product's development. Through our robust data and supportive evidence, we provide critical insights to help your combination product get to market on time.
At BD, we know that total system integration – where your drug formulation, primary container and secondary device require reliable combination to deliver dependable performance under real-life conditions – is critical to getting your drug to market quickly, efficiently and safely. To give you every advantage, we will help you anticipate and address a range of needs, from human factors aspects and use-related hazards to the complex regulatory environment.6,7
BD capabilities can support your combination development means.
Explore our products
- Analysis of BD sales from 2014 to 2018 [internal analysis]. Pont-de-Claix, FR: Becton Dickinson and Company; 2019
- Physioject™ post market surveillance quality statement [internal report]. Pont-de-Claix, FR: Becton, Dickinson and company; 2015
- BD External R&D Document: Design Control Evidence Intevia™ 1ml: PIR Number: 2014-0137 Document reference: ERD20200058
- BD design control procedure 103.107.350GP WW Product Design Control [internal document], Franklin Lakes, NJ, USA
- Market Research, “Challenges with System Integration" [external study], Franklin Lakes, NJ, USA: GLG 2019
- Zhaoyang Li & Rachael Easton (2018) Practical considerations in clinical strategy to support the development of injectable drug-device combination products for biologics, mAbs, 10:1, 18-33, DOI: 10.1080/19420862.2017.1392424 - ref-16128
- DeGrazio F, Paskiet D. Injectable Combination Product Development: Facilitating Risk-Based Assessments for Efficiency and Patient Centric Outcomes. J Pharm Sci. 2020 Jul;109(7):2101-2115. doi: 10.1016/j.xphs.2020.03.020. Epub 2020 Apr 6. PMID: 32272133 - ref-17453