BD maintains Product Security White Papers for its software-enabled products. The purpose of these documents is to provide details on how BD security and privacy practices have been applied and what our customers should know about maintaining security throughout the entire product lifecycle. Each white paper includes a Manufacturer Disclosure Statement for Medical Device Security (MDS2 attestation).
Product Security and Privacy Statement

- About Us
- Trust Center
- Bulletin and Patches
- Disclosure Process
- Helpful Resources
Cybersecurity is one of the most critical issues impacting the healthcare industry. At BD, we maintain an unwavering commitment to security by design, in use and through partnership. We strive to ensure our products, systems and customer environments maintain high security standards so our customers can focus on what matters most: caring for patients.
While we maintain robust security protocols, we also recognize that new security threats emerge daily, from attempts to compromise healthcare data to coordinated efforts to disrupt clinical workflows or manufacturing. We recognize that our customers cannot protect what they don’t know. That’s why we believe transparency and collaboration are essential. As we build a strong community of practice, working closely with our customers, industry regulators, and security researchers, we’re improving cybersecurity and resilience across the industry.
– Rob Suárez, Vice President and Chief Information Security Officer
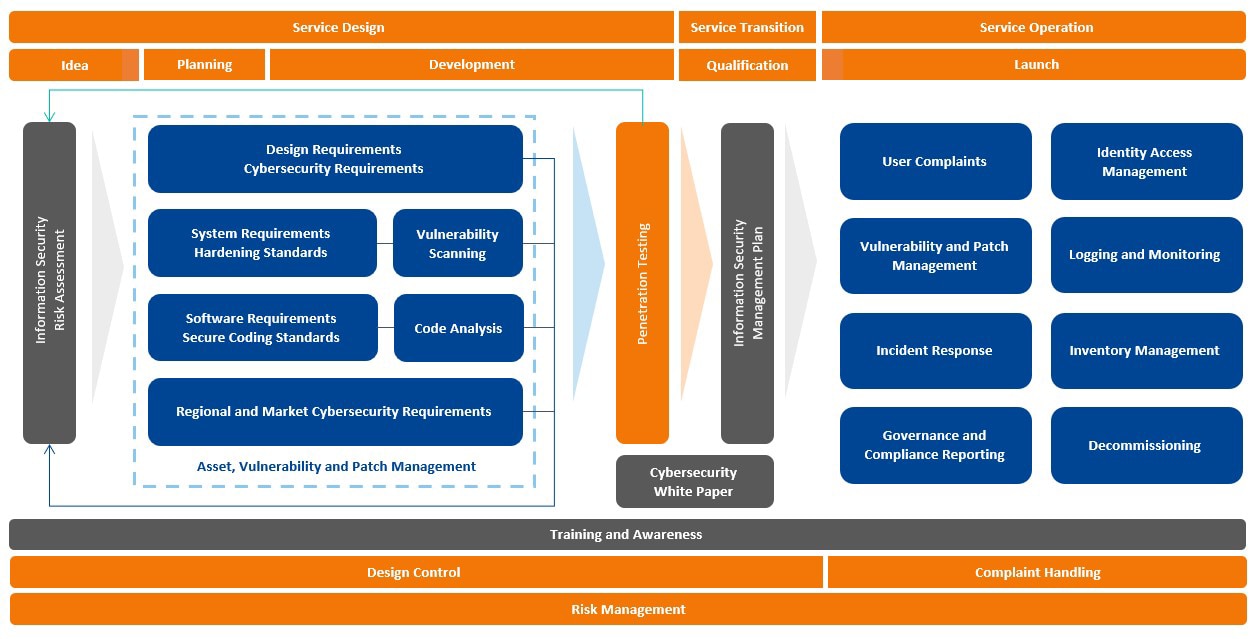
BD Cybersecurity Framework
BD utilizes a framework to incorporate cybersecurity into our processes for product design, manufacturing, customer support and enterprise systems. Our framework has been aligned to various industry work products including the HSCC Joint Security Plan, NIST Cybersecurity Framework, ISO 27001, UL 2900 and ISA 62443.
Access BD cybersecurity resources
BD recognizes the value to our customers of independent cybersecurity attestation. Each year a range of third-party audits are performed on BD products and internal cybersecurity controls. To demonstrate our commitment to product security and the protection of customer data, BD makes these industry recognized certifications and attestation reports available to customers.
SOC2+ reports and Product Security White Papers are restricted to existing BD customers and can be requested below. UL CAP certificates display the scope (product and version), validity period, and certifying UL Manager and can be downloaded below. Prospective customers that wish to obtain copies of SOC2+ reports or Product Security White Papers can request these from their sales representative following approval of a Confidential Disclosure Agreement (CDA). Select the documents you would like to access and use the icons at the bottom of the page to trigger the download or request.
Coordinated Vulnerability Disclosure
BD has established a routine practice of seeking, communicating, and addressing cybersecurity issues in a timely fashion. Vulnerability disclosure is an essential component to our approach to transparency by enabling customers to manage risk properly through awareness and guidance.
Find out how BD is advancing the world of health™ by driving collaboration across the industry, supporting our customers, and addressing the most pervasive cybersecurity challenges impacting the infrastructure of healthcare around the world.










